Introduction:
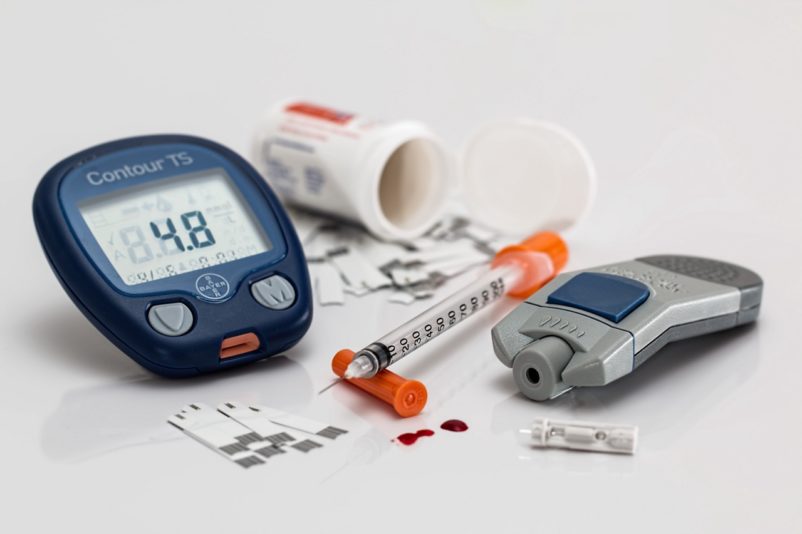
BS EN 13975:2003 specifies sampling procedure requirements for acceptance testing of finished in vitro diagnostic medical devices, which require EC verification by a notified body.
Two different provisions are addressed:
- Verification by testing attributes and/or variables on a statistical basis
- Verification by testing a homogeneous batch which has been defined by appropriate means of process validation and in-process control.
This European standard specifies requirements and criteria for testing procedures to establish and verify the homogeneity of processes and products. This standard is also applicable for drawing up sampling plans for finished products according to the requirements laid down for manufacturers’ product certification and production quality systems.
What is the BS EN 13975:2003?
The BS EN 13975:2003 is a Standard by which manufacturers of in vitro diagnostic medical devices have to fulfill criterion during their manufacturing process. It is a document which provides requirements to the manufacturers about sampling plans for their products.
What are the application and coverage of the Standard?
The BS EN 13975:2003 is applicable to in vitro diagnostic medical devices intended for use in the human body.
The sampling plan is to be used for prediction of the variation that might exist within the batch or lot that have been manufactured in a manufacturing facility. According to the Standard, the sampling plan and design of the sampling equipment are ones of the significant factors for acceptance of the manufactured batch or lot of products.
The standard is effective for all the manufacturers of in vitro diagnostic medical devices for using them for medical purposes. The Standard is mandatory for all the manufacturers which is classified as notified bodies for giving EC validation to the manufacturers of in vitro diagnostic medical devices.
The objective of the Standard is to ensure the quality for the manufacturer of in vitro diagnostic medical devices to continue their production process. The Standard specifies a criterion by which to verify the homogeneity of batches or lots of the finished products regarding the variability of key attributes and variables. The Standard contains no requirements on the methodology used within the process of process validation and in-process control.